2025 Postmarket Quality Benchmark Report
Industry data and insights on postmarket quality strategies
and challenges across medtech
Executive Summary
The 2025 Veeva MedTech Postmarket Quality Benchmark surveyed over 100 quality leaders across the industry to explore current postmarket quality processes and challenges.
The study shows that while organizations align on quality definitions and tactics, the lack of systems and tools remain a major barrier to proactive quality management. Half of medtech companies surveyed still rely on manual processes; and outdated systems are cited as the number one barrier, ranking 1.5 times higher than process or metric gaps.
Quality transformations are however gaining momentum, with 35% of organizations recently completing or undergoing a transformation and more than half planning to start soon. Advanced technology is seen as key to this progress, particularly in automation (75%), connectivity (62%), and reporting (61%). Companies already leveraging modern technology report improved workflow efficiency and fewer manual tasks.
This report outlines the key motivators for successful quality transformation, including improved governance, technology adoption, and proactive quality practices to reduce risk, improve compliance, and drive innovation.
Quality Transformations Gaining Momentum
Thirty-five percent of respondents indicate they are currently undergoing or have recently completed a postmarket quality transformation, while more than half (53%) are planning for transformation in the short-term.
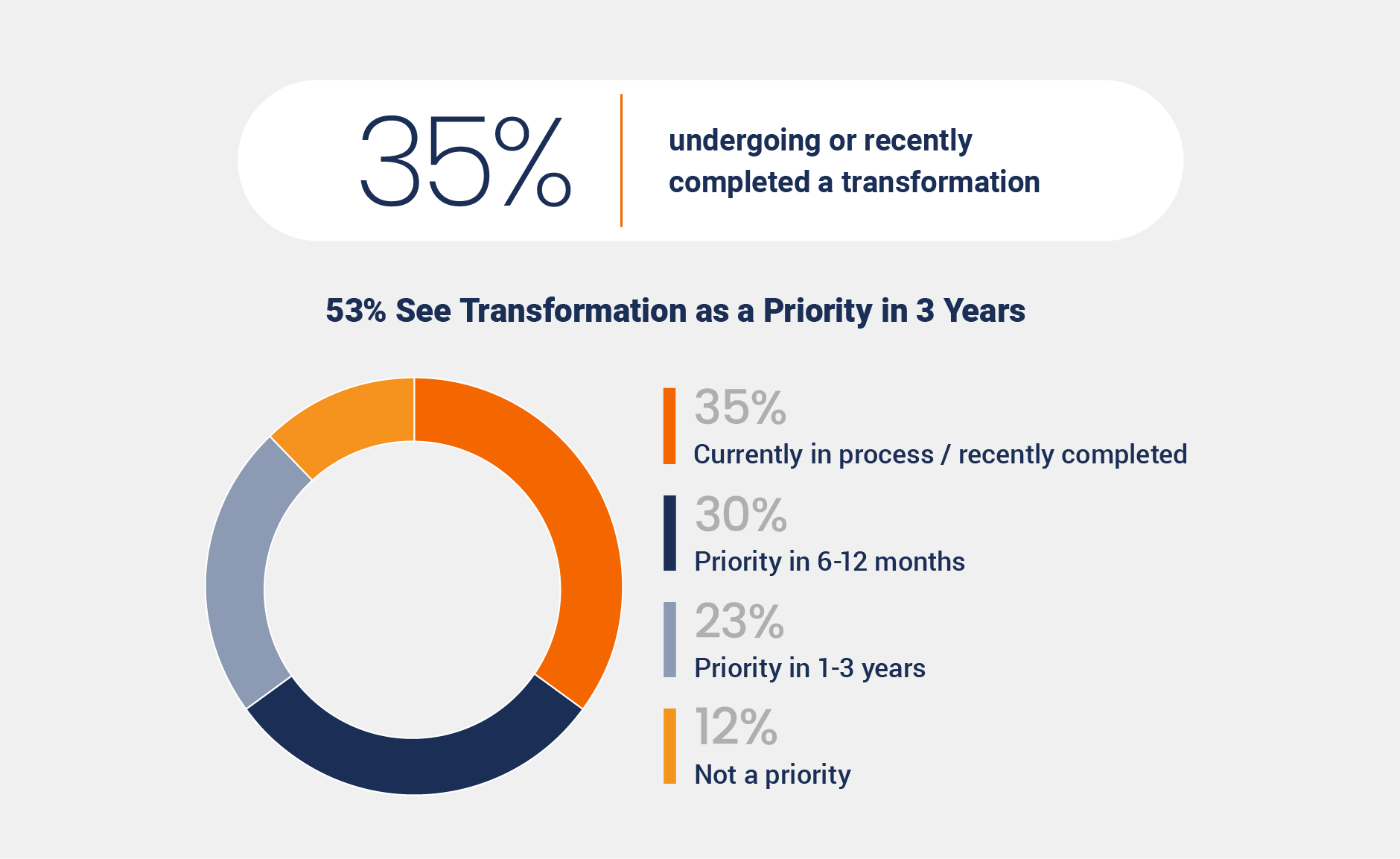
This momentum reflects the industry’s growing focus on proactive and risk-based quality management, with 38% of responding organizations citing this shift as a primary driver. Additionally, 26% point to the need to scale postmarket processes to support growth, underscoring the increasing emphasis on improving visibility, efficiency, and readiness for future demands.
Interestingly, while regulatory changes can act as catalysts for transformation, this factor ranked lower in priority, with just 10% of respondents citing evolving regulations. This finding suggests the industry is seeking to stay ahead of regulatory changes rather than react to them, underscoring the importance of readiness and proactive quality strategies.
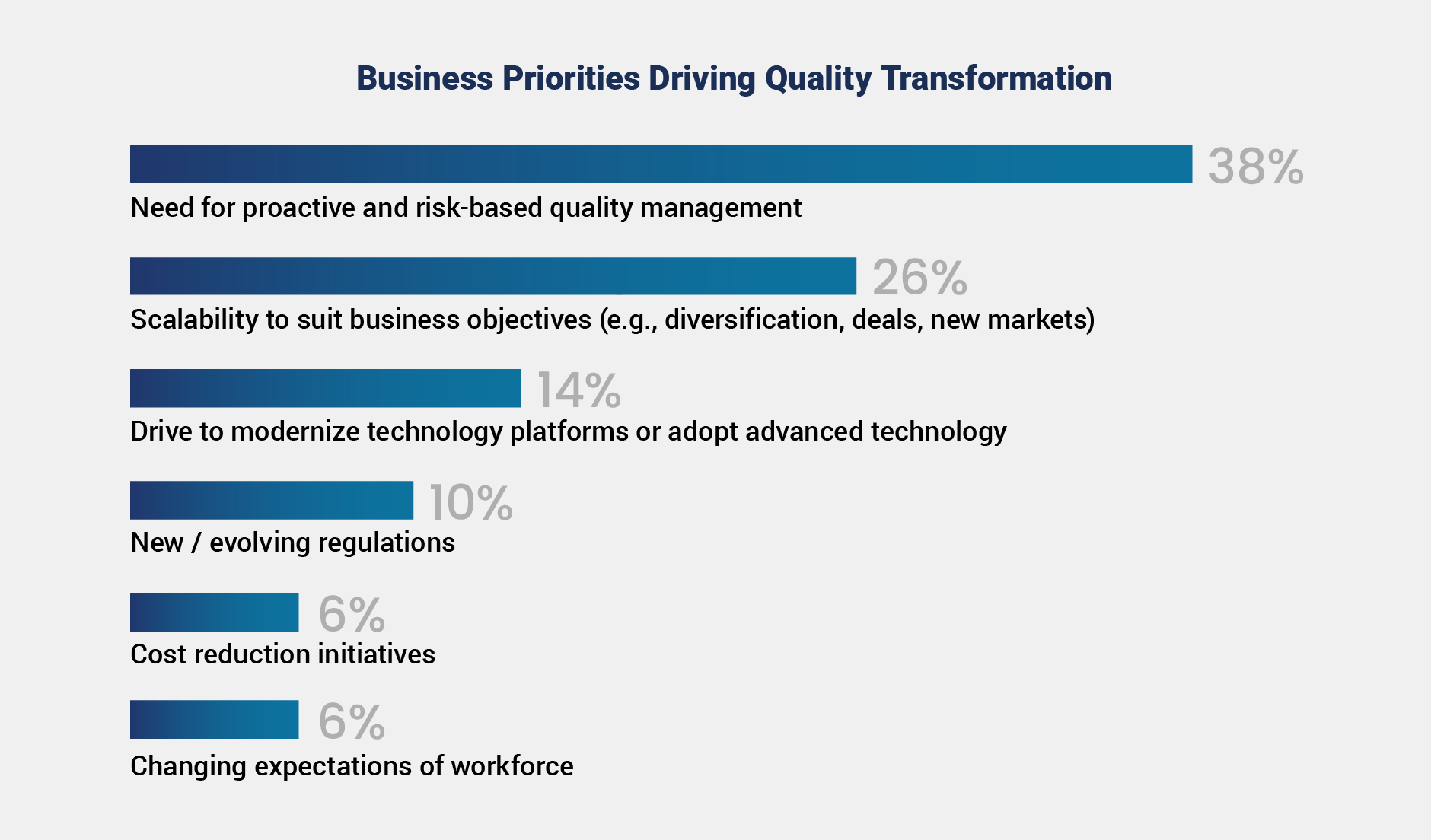
While technology is not the foundation of quality transformations, it plays a notable role, ranking third with 14% of respondents citing it as a priority for transformation. This reflects the industry’s recognition that modern tools can enable scalable processes, improve data visibility, and connect teams.
In fact, the study shows that 60% of surveyed organizations currently in transformation are already reliably using cloud-based technologies across the enterprise, with another 17% applying it in select areas.
Organizations advancing their transformation efforts have successfully leveraged technology to streamline processes and improve data access. By aligning technology adoption with clear priorities like risk-based thinking and scalable processes, organizations can drive lasting improvements in postmarket quality and business growth.
Quality Organization and Governance
A vast majority of medtech companies align on a core definition of postmarket quality focused on surveillance, reporting, and recalls, with 90% of respondents citing complaint handling as essential.
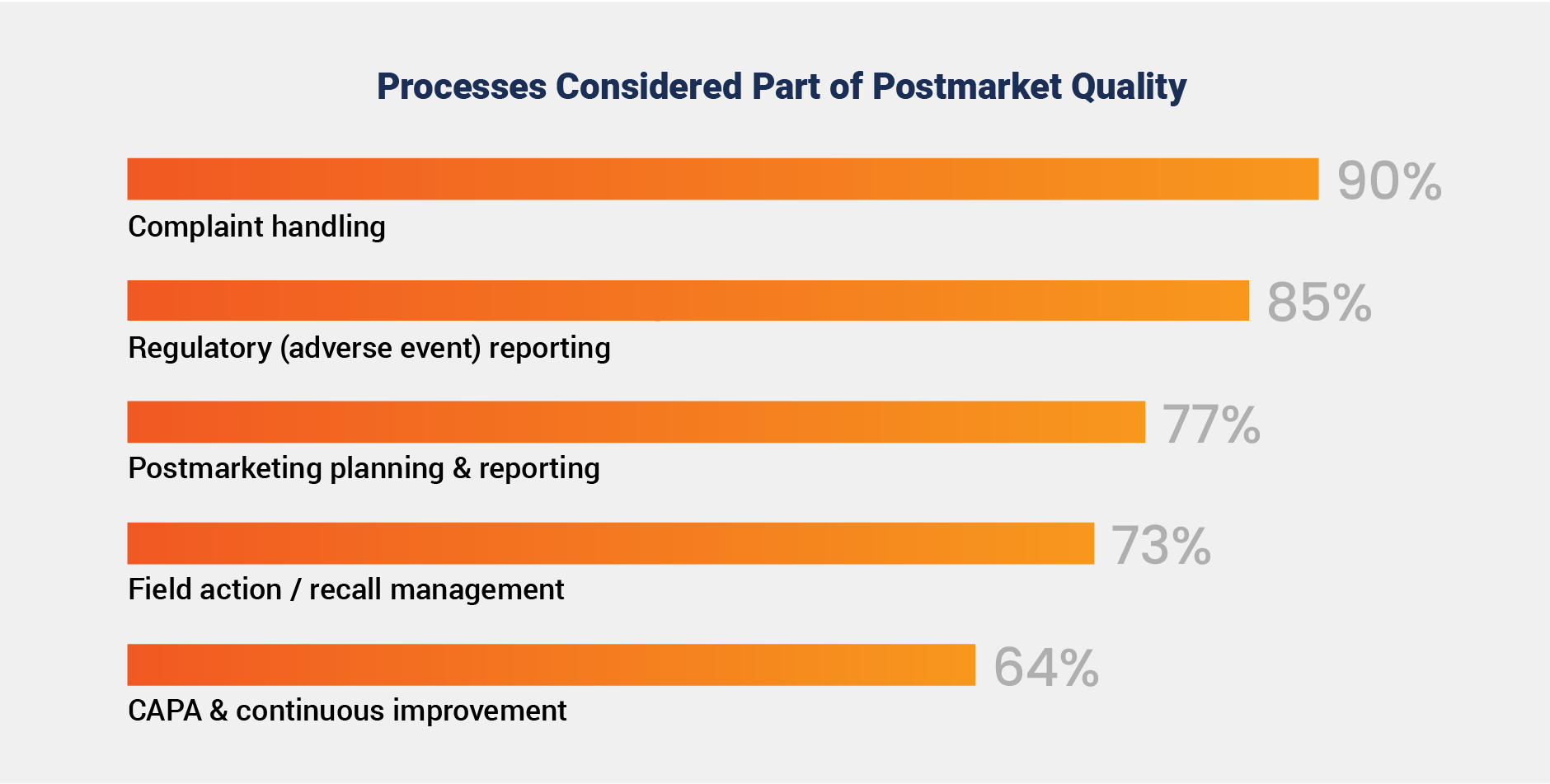
Notably, 64% also include CAPA and improvement initiatives, highlighting a recognition that postmarket quality is not only about issue resolution but also about driving long-term improvements and minimizing future risks. By actively linking CAPA back to the product lifecycle, organizations can drive a stronger feedback loop that informs design changes, enhances supplier management, and ultimately reduces the recurrence of quality issues.
The study reveals that 55% of responding organizations take a centralized approach to postmarket quality, enabling standardization, efficiency, and resource optimization. In contrast, 20% operate with decentralized models, offering flexibility but risking inconsistencies without robust systems.
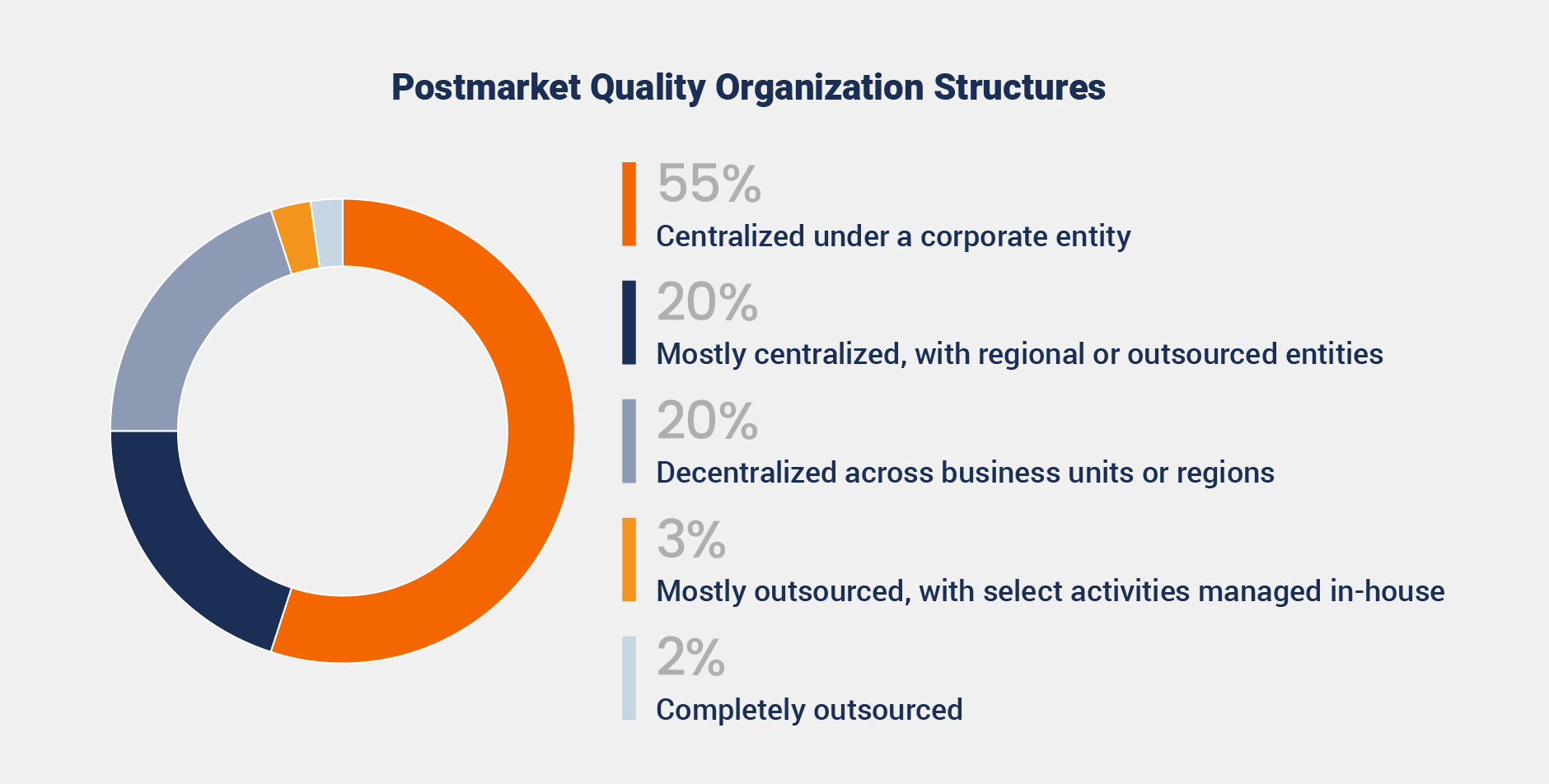
While centralization offers consistency, many organizations still rely on outdated technology. Among those with centralized or mostly centralized approaches, 68% of those surveyed are not using modern cloud-based systems.

This reliance on manual processes, mixed systems, and on-premise solutions limits efficiency, slows response times, and hinders the ability to act on quality insights. As a result, organizations may face delays in addressing product issues, which can lead to significant backlogs in complaint handling and failure to respond to patient safety concerns in a timely manner. This creates heightened compliance risks, particularly in an environment where delays can undermine brand reputation and customer trust. For example, companies reliant on outdated systems may struggle to manage complaint investigations efficiently, delaying critical actions such as product recalls or field corrections.
Cloud-based solutions can improve data traceability and collaboration, enabling faster decision-making and response times. By providing a single source of truth, these systems reduce transcription errors, improve data accuracy, and ensure teams have real time access to critical information. This visibility helps organizations address issues before they escalate, improving patient safety, reducing compliance risks, and protecting brand reputation. For example, streamlined complaint handling through cloud systems allows teams to respond quickly to emerging risks, reducing the chance of product recalls or noncompliance concerns.
For organizations still relying on outdated systems, adopting cloud solutions can be an effective step toward improving efficiency and ensuring quality processes keep pace with business demands
Knowledge Management Opportunities
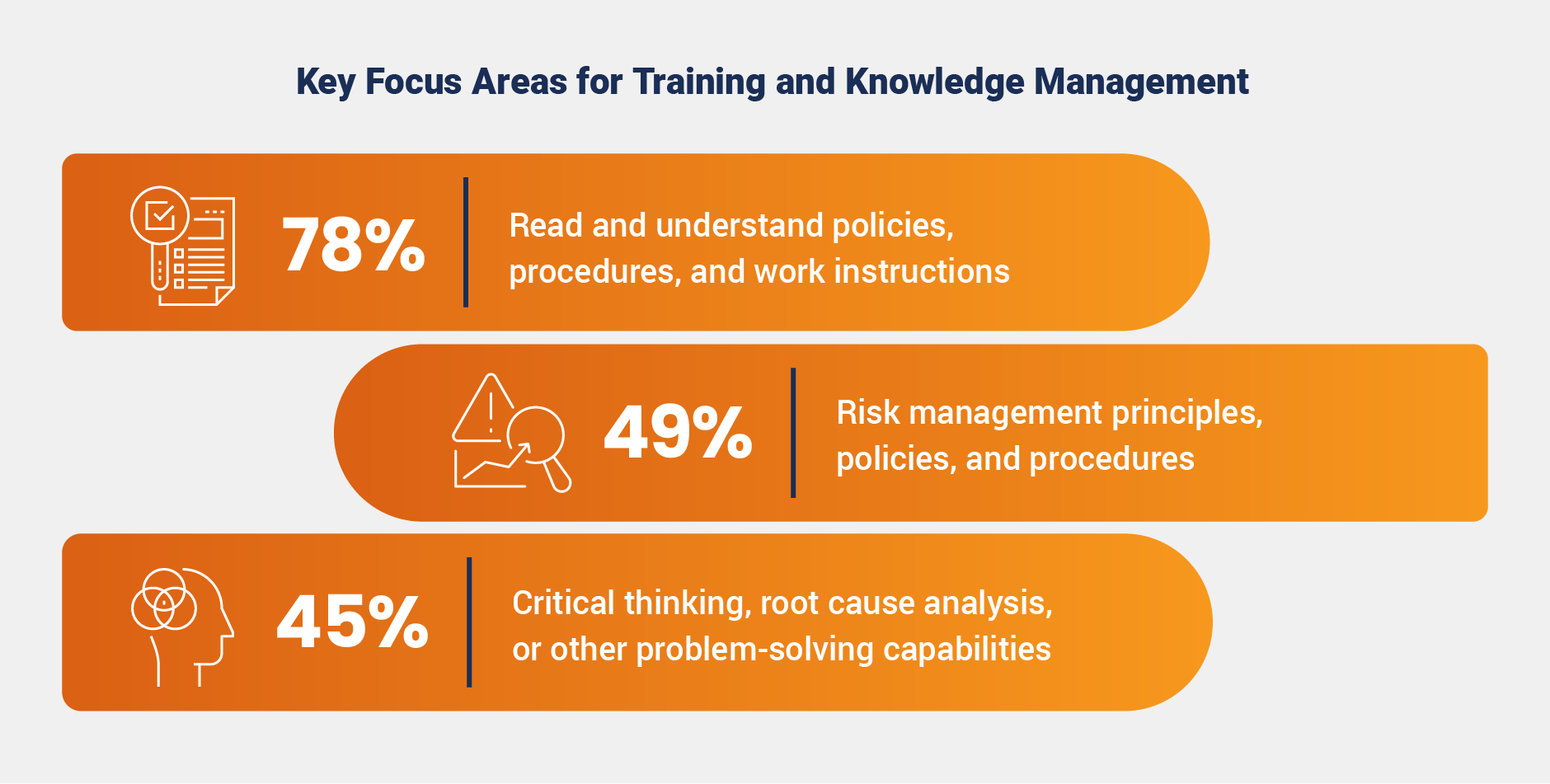
Training and knowledge management programs are heavily focused on reviewing policies, procedures, and work instructions, with nearly 80% of surveyed organizations prioritizing this area. This focus far outweighs other key areas such as risk management and problem-solving skills, revealing a potential gap in training programs. By placing greater emphasis on developing critical thinking and risk-based decision-making skills, organizations may better equip teams to manage postmarket challenges and improve product quality.
Medtech manufacturers may be recognizing this themselves, as a mere 19% of respondents feel their training programs are sufficient to support postmarket quality for future success, while 57% only ‘somewhat agree,’ further indicating room for improvement.

Among those recognizing gaps in their training programs, 70% have yet to adopt modern, cloud-based solutions. This highlights an opportunity to improve training methods by introducing interactive learning tactics and ensuring employees have access to real time updates and guidance.
As organizations look to elevate training programs, digital learning platforms present an opportunity to improve compliance and knowledge retention by delivering real-time updates and interactive learning experiences.
By centralizing training management, ensuring alignment with the most current procedures, and automating retraining based on changes, modern digital learning platforms can help reduce the risk of outdated procedures being followed while streamlining compliance efforts. These learning platforms also enable greater consistency and visibility across teams, allowing quality organizations to scale training more effectively and focus on building capabilities—like risk management and problem-solving—that are critical to long-term postmarket success.
Shift Towards Proactive Quality Management
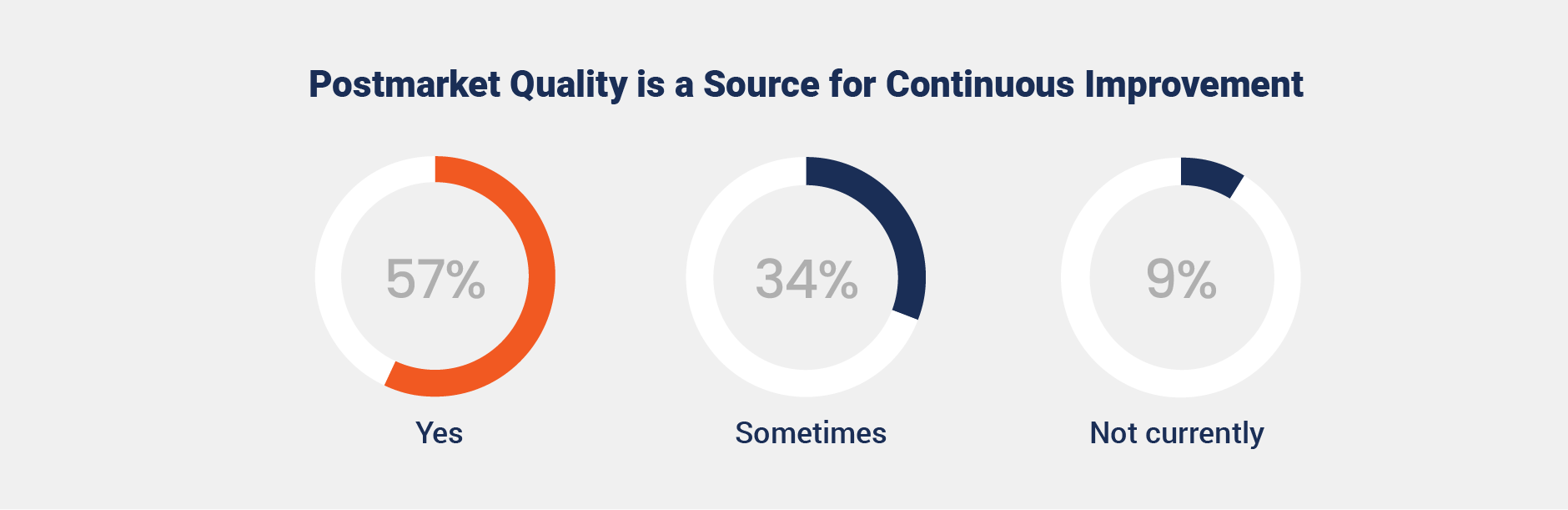
Organizations increasingly recognize postmarket quality as a strategic driver of continuous improvement and risk mitigation. More than half of respondents (57%) report using postmarket quality insights to refine processes, enhance compliance, and identify opportunities for innovation.
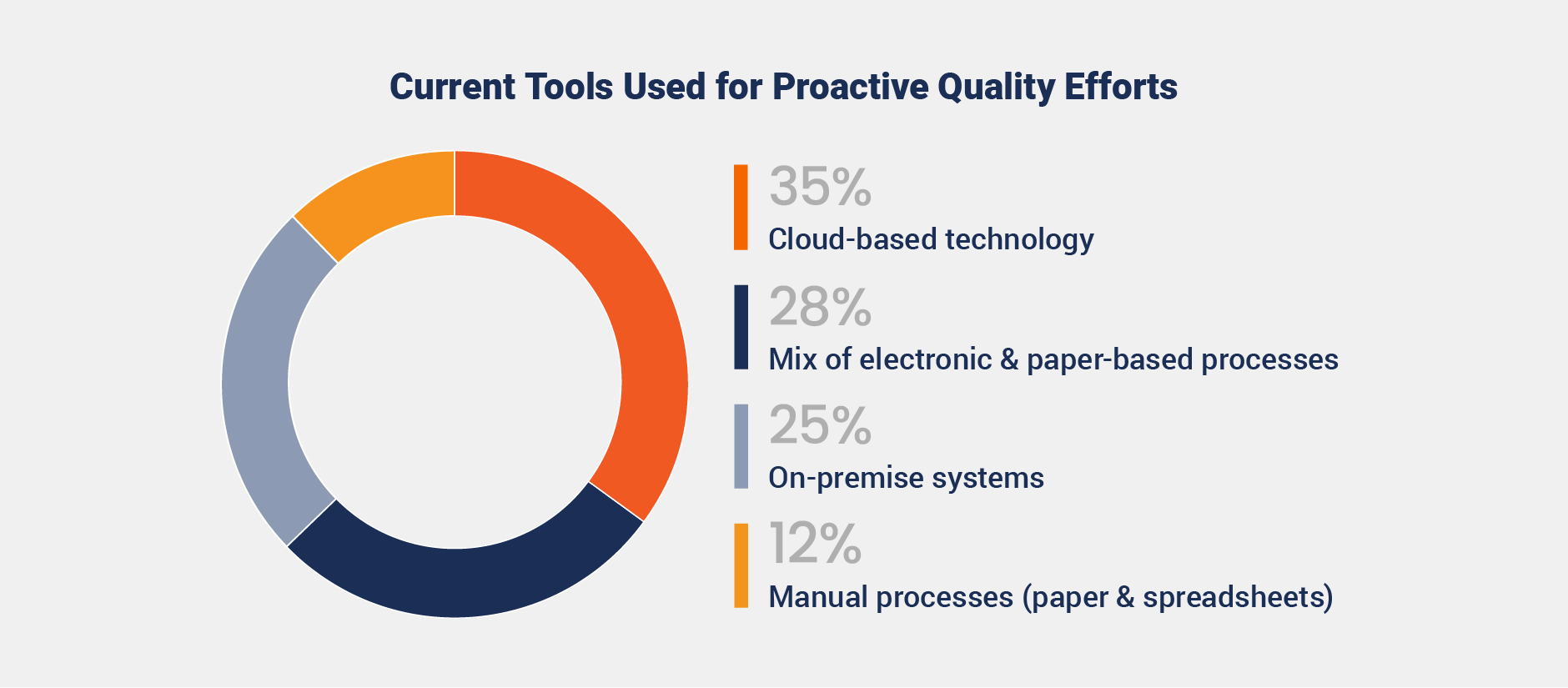
However, fragmented systems are limiting progress. While 35% of organizations use cloud-based technology to support proactive quality, others rely on less efficient tools such as on-premise systems (25%) or a mix of electronic and paper-based processes (28%)
Even as device and diagnostic companies aim to be more proactive, many are still relying on outdated or disconnected systems that limit their ability to access insights and act on them effectively. This is reflected in the fact that the lack of systems and tools to access and analyze data was cited as the most significant challenge, ranking nearly 1.5 times higher than gaps in processes, definitions, or metrics.

This highlights the need for modern solutions that enable faster access to postmarket insights and supports proactive decision-making. Without these solutions, organizations risk working with incomplete or delayed data, making it harder to identify trends, address risks, and implement preventive measures.
For example, a medtech relying on disconnected systems may struggle to quickly trace the root cause of recurring complaints. Without centralized data, teams can waste valuable time piecing together information from separate sources, delaying corrective actions and increasing compliance risks. Organizations using modern solutions, however, can connect postmarket data with design, manufacturing, and supplier insights to identify trends faster, implement targeted improvements, and improve product quality.
Investing in systems that centralize data, enhance visibility, and provide real-time insights is key to transforming postmarket quality from a reactive process into a strategic advantage that drives faster issue resolution, strengthens product reliability, and improves patient safety.
Advanced Technology: Adoption vs. Potential
While many medtech organizations are focused on quality transformations, technology adoption to support these efforts remains limited. Over half (51%) of responding companies still rely on manual or partially manual processes to manage postmarket quality.

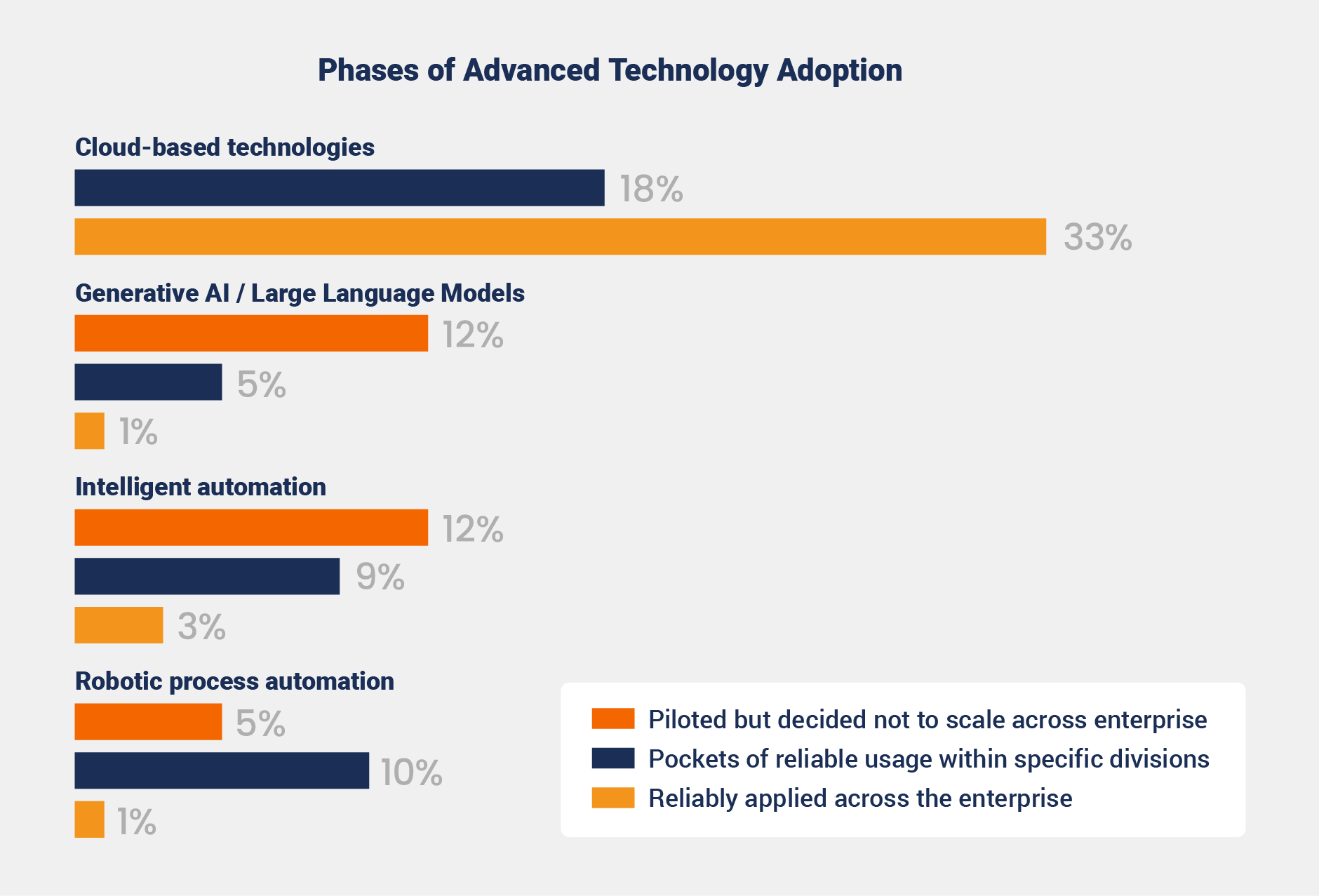
Technology adoption remains widely inconsistent across medtech manufacturers. Cloud-based technologies show the strongest traction, with 33% of surveyed organizations reporting reliable enterprise-wide use and 18% noting limited adoption.
Smaller device and diagnostics companies are adopting cloud solutions at higher rates, with 32% of respondents reporting reliable use compared to 21% of those at larger organizations. This reflects the complexity larger companies face when transitioning from manual or legacy systems, making it more challenging to implement modern solutions at scale.
Emerging innovations like generative AI, intelligent automation, and robotic process automation are still in the early stages, with limited adoption across the industry.
Despite limited adoption, there is strong optimism about the potential of advanced technologies to improve postmarket quality. Respondents identified automation to improve timeliness and efficiency (75%), connections with other QMS processes (62%), and advanced reporting to support data-driven decisions (61%) as top areas for impact. Data indicates less focus on use cases of error-checking and improving business logic to streamline workflows.
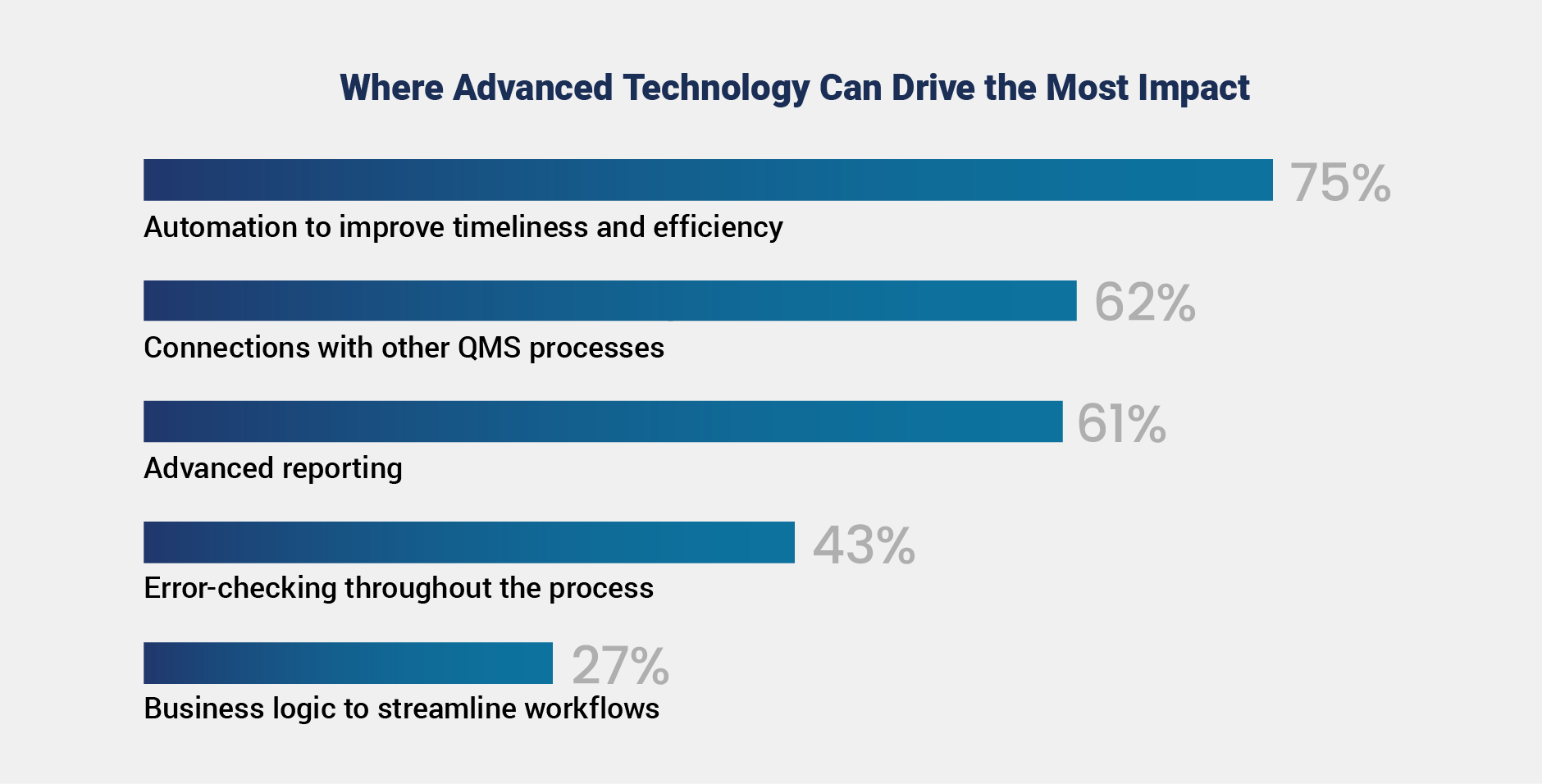
As organizations advance their quality transformation efforts, technology can be a powerful enabler of both large-scale change and targeted improvements. For companies pursuing broad transformation, technology can enhance outcomes by connecting systems, streamlining workflows, and improving data access. Medtech organizations earlier in their journey or with a gradual adopting approach can still achieve meaningful results by focusing on high-impact areas.
To maximize the value of technology investments for quality, organizations should:
- Prioritize clear objectives: Define specific outcomes like improved complaint resolution times, faster CAPA closure, or improved audit readiness to guide implementation efforts.
- Start with high-impact areas: Focus initial adoption on quality processes that are heavily manual, fragmented, or prone to compliance risk to deliver immediate value.
- Ensure cross-functional alignment: Engage quality, IT, and business teams early in the process to help align technology goals with broader transformation priorities.
- Leverage best practices for implementation: Learning from successful implementations can improve adoption outcomes and ensure long-term success. One medtech company’s experience implementing a cloud-based QMS highlights the importance of setting clear objectives, engaging cross-functional teams, and taking a phased approach to ensure lasting value.
Aligning technology adoption with both long-term goals and immediate priorities helps reduce complexity, demonstrate value early, and expand adoption over time to drive stronger postmarket quality outcomes and business growth.
Conclusion
The 2025 Veeva MedTech Postmarket Quality study shows that while medtech organizations align on postmarket quality processes, outdated systems remain a barrier to proactive quality management. Although many companies are actively pursuing transformation, success hinges on modernizing technology to improve data access, connectivity, and insights.
To achieve meaningful progress, organizations should address gaps in infrastructure, align technology adoption with strategic goals, and enable proactive quality practices. Investing in modern systems can help streamline postmarket quality, improve data visibility, and enhance training to move organizations from reactive issue management to proactive risk prevention and innovation.
By replacing manual processes and legacy systems with scalable solutions, device and diagnostics companies can improve postmarket quality, leverage data to spot trends, and foster a stronger culture of quality across the product lifecycle.
Achieving these outcomes requires thoughtful planning, clear objectives, and strategic technology investments. With the right approach, medtech organizations can build more resilient quality systems that improve product quality, maintain compliance, and drive long-term business success.
Discover how Veeva Quality Cloud enables proactive quality management, accelerating technology adoption and transformation efforts to enhance patient safety, minimize risk, and drive market success.
Survey Methods & Respondents
The study surveyed more than 100 quality management leaders at medical device and diagnostic companies globally. The survey included 12 required core questions about postmarket quality management, alongside additional demographic questions. Completion of the survey was voluntary and confidential.